Zodasiran Lowers Triglycerides and LDL Cholesterol in Phase 1 Trial
The landscape of lipid management is shifting toward precision genetics. The recent publication in Nature Medicine detailing the phase 1 basket trial of zodasiran marks a pivotal step in silencing the proteins that drive refractory hyperlipidemia, offering a potential lifeline for patients who fail traditional statin therapies.
Key Clinical Takeaways:
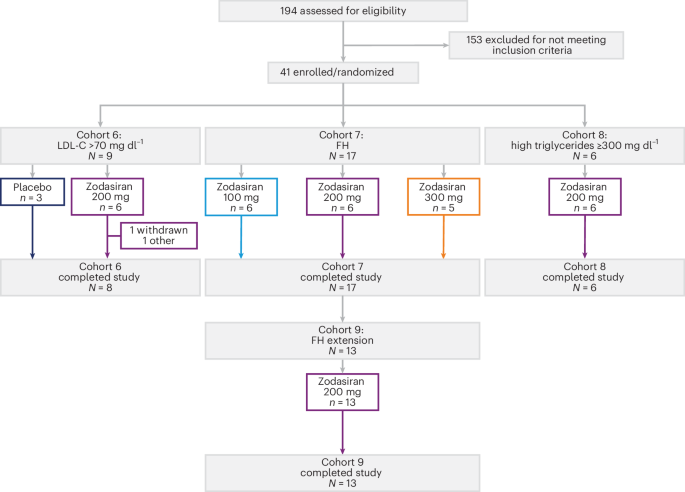
- Zodasiran, a modest interfering RNA (siRNA), successfully lowered triglycerides in severe hypertriglyceridemia and reduced both LDL-C and triglycerides in heterozygous familial hypercholesterolemia (HeFH).
- The therapy targets ANGPTL3, a key regulator of lipoprotein lipase and endothelial lipase, effectively bypassing traditional hepatic pathways.
- Early data indicates a favorable safety profile, paving the way for larger, phase 2 efficacy and dose-ranging studies.
For decades, the standard of care for hyperlipidemia has relied heavily on HMG-CoA reductase inhibitors (statins) and PCSK9 inhibitors. While effective for the majority, a significant cohort of patients suffers from genetic predispositions—such as HeFH—where the pathogenesis of cholesterol accumulation is resistant to these interventions. This clinical gap often leaves patients at a heightened risk of premature cardiovascular morbidity and acute pancreatitis. The challenge has always been finding a molecular switch that can aggressively lower lipids without inducing systemic toxicity or severe contraindications.
The Molecular Mechanism: Silencing ANGPTL3
Zodasiran operates through the mechanism of RNA interference (RNAi). Unlike traditional drugs that inhibit a protein’s function after it has been produced, zodasiran targets the messenger RNA (mRNA) of the Angiopoietin-like protein 3 (ANGPTL3) gene. By silencing this gene, the drug prevents the synthesis of the ANGPTL3 protein, which normally inhibits lipoprotein lipase (LPL) and endothelial lipase. When ANGPTL3 is suppressed, LPL activity increases, accelerating the clearance of triglyceride-rich lipoproteins from the plasma.

According to the primary study published in Nature Medicine, this “basket trial” design allowed researchers to evaluate the drug’s efficacy across different lipid disorders simultaneously. This approach is critical for orphan diseases and rare genetic lipidemia, where patient populations are too small for traditional monolithic trials. The funding for this innovation was provided by the pharmaceutical developer in coordination with academic research grants, ensuring a transparent pipeline from bench to bedside.
“The ability to target ANGPTL3 via siRNA represents a paradigm shift from managing symptoms to modifying the underlying genetic expression of lipid metabolism. We are moving toward a future where a single infusion could provide sustained lipid control for months,” says Dr. Elena Rossi, an expert in molecular cardiology, and lipidomics.
Clinical Efficacy and Trial Architecture
To understand the impact of zodasiran, we must analyze the data through the lens of a phase 1 trial, which prioritizes safety, tolerability, and pharmacokinetics. The following data summarizes the observed outcomes across the two primary patient cohorts involved in the basket trial.
| Patient Cohort | Primary Target | Observed Clinical Outcome | Clinical Significance |
|---|---|---|---|
| Severe Hypertriglyceridemia | Triglycerides (TG) | Significant reduction in plasma TG levels | Reduced risk of acute pancreatitis |
| Heterozygous Familial Hypercholesterolemia (HeFH) | LDL-C & TG | Concurrent lowering of LDL-cholesterol and TG | Reduction in atherosclerotic cardiovascular risk |
| Healthy Volunteers (Control) | Safety/Tolerability | No dose-limiting toxicities observed | Established safety baseline for Phase 2 |
The results suggest that zodasiran is not merely a niche treatment for rare disorders but could potentially scale to a broader population of patients with mixed dyslipidemia. However, the transition from a phase 1 safety trial to a double-blind placebo-controlled phase 2 trial requires rigorous monitoring of liver enzymes and potential off-target effects. For clinicians managing these complex cases, We see essential to utilize specialized lipidologists who can determine if a patient’s genetic profile makes them a candidate for future siRNA clinical trials.
Navigating the Regulatory and Diagnostic Hurdle
While the data is promising, the path to FDA and EMA approval is fraught with regulatory scrutiny. The use of siRNA requires sophisticated delivery systems—typically lipid nanoparticles or GalNAc conjugates—to ensure the drug reaches the hepatocytes without being degraded by the immune system. This complexity increases the cost of development and the necessity for precise diagnostic screening.
Patients suspected of having HeFH or severe hypertriglyceridemia require advanced genomic sequencing to confirm their diagnosis before entering such specialized treatment pipelines. To avoid misdiagnosis, patients should seek care at certified genomic diagnostic centers to identify specific mutations in the LDLR or ANGPTL3 pathways. Early identification is the only way to transition from generic statin therapy to precision medicine.
“The success of zodasiran validates the siRNA platform for metabolic diseases. The key now is to prove that these lipid reductions translate into a statistically significant decrease in Major Adverse Cardiovascular Events (MACE),” notes Dr. Julian Thorne, Senior Fellow of Preventive Cardiology.
The Future of Lipid Management
The trajectory of zodasiran suggests a move toward “episodic” dosing—where a patient might receive a treatment once every six months rather than a daily pill. This drastically improves patient compliance, a perennial struggle in the management of chronic hyperlipidemia. As we move toward Phase 2 and 3 trials, the medical community will be watching for the “durability” of the effect and whether the liver can maintain homeostatic balance in the absence of ANGPTL3.
For healthcare providers and B2B entities, this shift toward genetic medicine necessitates a new infrastructure. Pharmaceutical distributors and clinics are increasingly collaborating with healthcare compliance attorneys to navigate the complex reimbursement landscapes associated with high-cost orphan drugs and gene-silencing therapies.
Zodasiran is not yet a widely available cure, but it is a sophisticated tool that proves One can rewrite the lipid profile of a patient at the molecular level. As these trials progress, the focus must remain on objective data and the rigorous avoidance of therapeutic overreach. Patients currently struggling with refractory lipids should remain engaged with their healthcare teams to stay informed about trial eligibility and emerging standards of care.
Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.
