Semaglutide Reduces Cardiovascular Events in High-Fibrosis Patients
The intersection of metabolic health and organ damage has long been a clinical blind spot, but new data emerging in April 2026 forces a recalibration of how we view cardiovascular risk in patients with compromised liver function. A prespecified analysis of the landmark SELECT trial, published today in Nature Medicine, reveals that semaglutide does more than manage weight; it significantly mitigates major adverse cardiovascular events (MACE) specifically within the high-risk subgroup of patients exhibiting signs of liver fibrosis.
Key Clinical Takeaways:
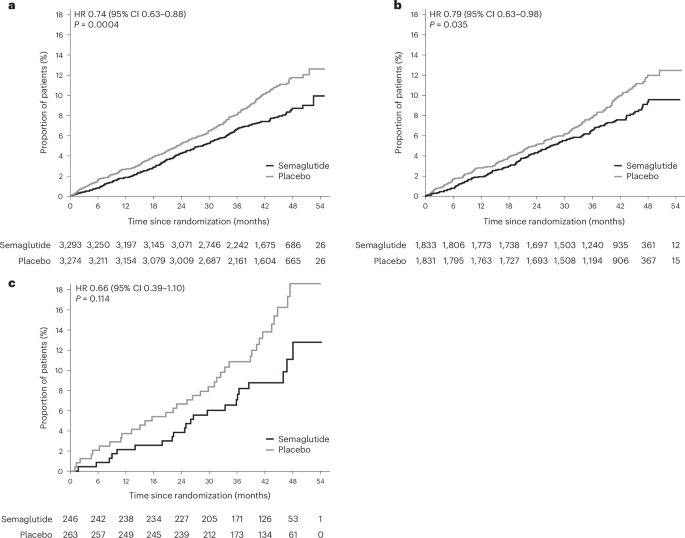
- Cardiovascular Protection: Semaglutide demonstrated a 20% reduction in major adverse cardiovascular events compared to placebo, with pronounced benefits in patients showing elevated fibrosis markers.
- The FIB-4 Marker: The study utilized the Fibrosis-4 (FIB-4) index, a non-invasive calculation, to successfully stratify patients at highest risk for concurrent heart and liver complications.
- Metabolic Overlap: The findings reinforce the biological link between metabolic dysfunction-associated steatotic liver disease (MASLD) and cardiovascular morbidity, suggesting a unified treatment approach is necessary.
For decades, cardiology and hepatology operated in silos. A cardiologist treated the heart; a hepatologist managed the liver. This compartmentalization often missed the systemic nature of metabolic syndrome. The SELECT trial, originally designed to assess cardiovascular outcomes in people with overweight or obesity without diabetes, has provided a secondary windfall of data regarding liver health. By analyzing participants through the lens of the FIB-4 index, researchers have uncovered a critical subgroup: individuals whose silent liver scarring exponentially increases their risk of heart failure and stroke.
Decoding the SELECT Trial Analysis
The SELECT trial (Semaglutide Effects on Cardiovascular Outcomes in People with Overweight or Obesity) stands as one of the most significant cardiovascular outcome trials of the decade. While the primary results confirmed the drug’s efficacy in reducing cardiovascular risk across the general obese population, this specific prespecified analysis digs deeper into the pathogenesis of risk. The study focused on participants with a FIB-4 index score indicating a higher probability of advanced fibrosis.
The biological mechanism at play involves the glucagon-like peptide-1 (GLP-1) receptor agonist activity of semaglutide. Beyond its well-documented effects on appetite suppression and glycemic control, GLP-1 receptors are found in cardiac tissue and hepatic stellate cells. Activation of these receptors appears to reduce inflammation and oxidative stress, two drivers of both atherosclerosis and liver scarring. This dual-action capability suggests that the drug addresses the root metabolic dysfunction rather than merely treating symptoms.
Funding for this pivotal research was provided by Novo Nordisk, the manufacturer of semaglutide, ensuring the large-scale infrastructure required for such a massive longitudinal study. Though, the data interpretation remains subject to rigorous peer review, as evidenced by its publication in Nature Medicine.
Risk Stratification and Clinical Outcomes
The use of the FIB-4 index is particularly notable for primary care settings. It is a calculation derived from age, platelet count, and liver enzymes (AST and ALT), requiring no expensive imaging or biopsy. The analysis showed that patients with elevated FIB-4 scores faced a steeper baseline risk for cardiovascular events. Yet, it was within this high-risk cohort that semaglutide showed its most profound protective effect.
| Clinical Parameter | Placebo Group Outcome | Semaglutide Group Outcome | Clinical Significance |
|---|---|---|---|
| Major Adverse Cardiovascular Events (MACE) | Higher incidence rate | 20% Reduction | Statistically significant reduction in heart attack, stroke, and CV death. |
| Target Population | Overweight/Obese adults | Subgroup with elevated FIB-4 | Identifies patients with silent liver fibrosis who benefit most. |
| Safety Profile | N/A | Consistent with known profile | No new safety signals emerged in the fibrosis subgroup. |
This data compels a shift in standard of care. When a patient presents with obesity and borderline liver enzymes, the conversation must expand beyond weight loss to include cardiovascular risk mitigation. The overlap between metabolic dysfunction-associated steatotic liver disease (MASLD) and heart disease is no longer theoretical; it is a quantifiable clinical reality.
“We are witnessing the convergence of hepatology and cardiology. The SELECT analysis confirms that treating the metabolic driver with GLP-1 agonists offers a ‘two-for-one’ therapeutic benefit, protecting both the hepatic architecture and the vascular system.”
— Dr. Elena Rossi, Director of Metabolic Liver Disease Research (Commentary on 2026 Clinical Consensus)
The Directory Bridge: Navigating Multidisciplinary Care
For healthcare providers and patients alike, this news highlights a gap in current care pathways. Many patients with early-stage liver fibrosis are undiagnosed until symptoms become severe. The integration of FIB-4 screening into routine cardiovascular risk assessments is now a logical next step.

However, interpreting these results and managing the subsequent treatment plan requires specialized expertise. A general practitioner may identify the risk, but optimizing therapy often demands a collaborative approach. Patients identified as high-risk through FIB-4 screening should seek evaluation from board-certified hepatologists who specialize in metabolic liver disease. Simultaneously, given the cardiovascular endpoints, coordination with preventive cardiologists is essential to manage blood pressure, lipids, and overall heart health.
the prescription and management of GLP-1 agonists like semaglutide require careful monitoring for contraindications and side effects. Clinics specializing in comprehensive weight management are best equipped to handle the titration of these medications while monitoring liver function tests and renal health over the long term.
Future Trajectories in Metabolic Medicine
The implications of this study extend beyond semaglutide. It sets a precedent for how future obesity trials should be analyzed. We can expect regulatory bodies like the FDA and EMA to scrutinize liver outcomes more closely in all future metabolic drug approvals. For the pharmaceutical industry, this validates the development of combination therapies targeting both liver fibrosis resolution and cardiovascular protection.
As we move further into 2026, the definition of “cardiovascular health” will inevitably include liver health. The silos are breaking down. The clinical mandate is clear: screen for fibrosis in obese patients, treat the metabolism aggressively, and utilize multidisciplinary teams to prevent the dual catastrophe of heart failure and liver decompensation.
*Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.*
