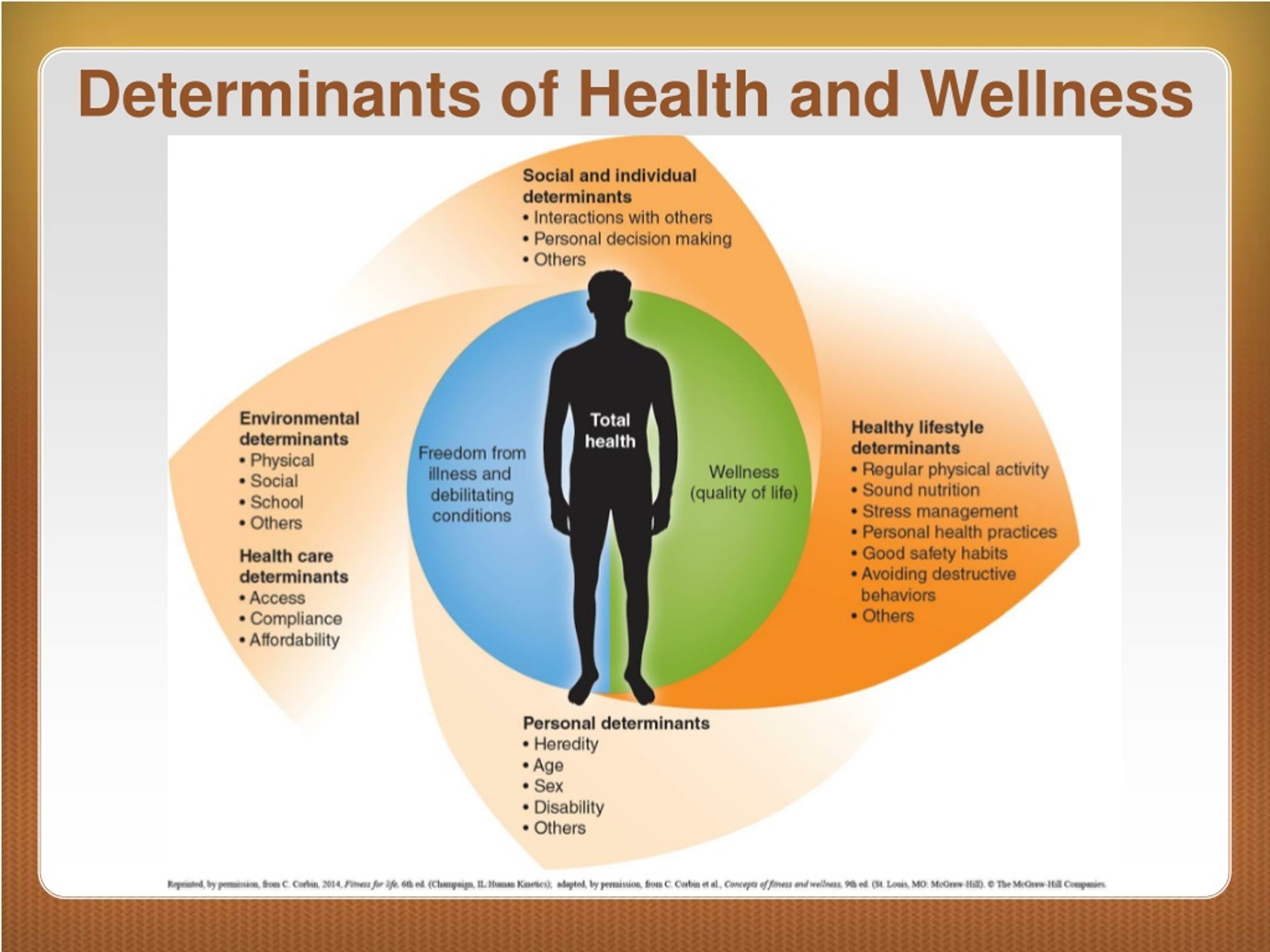
Quality Health Information: A Fundamental Determinant of Health
Access to accurate medical data is not merely a convenience. it is a biological imperative. When patients cannot distinguish between peer-reviewed evidence and anecdotal noise, morbidity rates climb. A landmark study published today in Nature Medicine confirms that quality health information functions as a fundamental determinant of health, equivalent in weight to sanitation or nutrition. As we navigate the complex landscape of 2026, the gap between data generation and patient comprehension remains the most critical vulnerability in modern healthcare infrastructure.
- Key Clinical Takeaways:
- Information asymmetry directly correlates with increased patient mortality and decreased adherence to standard of care protocols.
- Rigorous clinical trial transparency (Phase 1 through Phase 3) is essential for validating therapeutic safety before public dissemination.
- Healthcare systems must integrate certified patient advocacy and compliance resources to bridge the literacy gap.
The longitudinal analysis published in Nature Medicine on April 2, 2026, establishes a causal link between health literacy and clinical outcomes. The research indicates that populations with direct access to verified clinical data demonstrate significantly lower rates of preventable hospitalization. This finding shifts the paradigm of public health strategy. Instead of viewing information as a secondary support tool, medical governance bodies must treat data accessibility as a primary intervention. The study, funded by the Global Health Equity Consortium, underscores that without clear communication channels, even the most advanced investigational medicinal products (IMPs) fail to reach their potential impact.
Understanding where quality information originates is vital for assessing its reliability. The drug development process begins long before a treatment reaches a pharmacy shelf. According to the U.S. Food and Drug Administration, clinical research represents Step 3 in the development process, following extensive preclinical research. This phase answers basic questions about safety and biological activity. However, the complexity of this data often creates a barrier. Phase 1 clinical trials, for instance, focus primarily on safety and dosage in small groups, whereas Phase 3 trials test efficacy in larger populations. When patients encounter headlines about a drug in Phase 1, they often misunderstand the preliminary nature of the findings. This confusion highlights the need for certified medical communicators who can translate regulatory milestones into actionable patient knowledge.
The Health Research Authority in the United Kingdom emphasizes that all investigational medicinal products travel through a series of trials designed to protect patient interests. Yet, the sheer volume of technical documentation can overwhelm both providers, and patients. The National Heart, Lung, and Blood Institute notes that clinical trials are the first step in trying an investigational drug in humans to test safety and effectiveness. When this information remains siloed within academic journals or regulatory databases, it ceases to function as a public health tool. The Nature Medicine report argues that democratizing this data requires structural changes within healthcare delivery systems.
“Transparency without comprehension is noise. We must build infrastructure that not only releases data but ensures it is understood by the communities most at risk. This is where clinical governance meets social responsibility.”
— Dr. Elena Rosetti, Senior Epidemiologist, Global Health Equity Consortium
Bridging this gap requires active intervention from healthcare organizations. Clinics and hospital systems must audit their patient education materials against current regulatory standards. For healthcare providers navigating the sudden shift in data dissemination guidelines, retaining healthcare compliance attorneys is becoming standard practice to avoid operational bottlenecks. Legal experts ensure that patient communication aligns with FDA and EMA guidance, preventing the accidental dissemination of unverified claims that could jeopardize patient safety or regulatory standing. This legal backbone supports the clinical mission, ensuring that the information flowing to patients is both accurate and compliant.
the role of specialized diagnostic centers cannot be overstated. When patients encounter conflicting information online, they require a trusted physical location to verify their health status. For individuals experiencing persistent symptoms despite following general wellness advice, adjusting care protocols is urgent. It is highly recommended to consult with vetted accredited diagnostic centers to explore emerging therapies grounded in verified trial data. These centers serve as the filter between raw clinical data and personalized medical advice, ensuring that the determinant of health remains positive rather than confusing.
The semantic landscape of modern medicine is dense with terms like pathogenesis, contraindications, and double-blind placebo-controlled methodologies. While necessary for scientific precision, these terms create friction for the layperson. The Nature Medicine study suggests that reducing this linguistic friction is a clinical imperative. Health systems that invest in plain-language summaries of complex trial data see higher engagement rates and better adherence to treatment plans. This approach aligns with the FDA’s goal of ensuring patients understand the drug development process, specifically how preclinical research transitions into human clinical research.
Looking toward the future trajectory of this research, the integration of artificial intelligence in summarizing clinical trials offers promise but introduces new risks regarding accuracy. Human oversight remains non-negotiable. The medical community must prioritize the training of professionals who can interpret high-level data for public consumption. As we move further into 2026, the distinction between a healthy population and an at-risk one will increasingly depend on who holds the keys to understanding medical evidence. Providers who prioritize this informational hygiene will lead the next era of preventive care.
*Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.*
