PANGEA-SMM: A Novel Prediction Model for Smoldering Multiple Myeloma Progression Using Dynamic Biomarkers and Large International Cohorts
BOSTON – A latest predictive model for smoldering multiple myeloma (SMM), developed by researchers at Dana-Farber Cancer Institute (DFCI), significantly improves risk stratification compared to existing methods, according to a study published today in Nature Medicine. The model, dubbed PANGEA-SMM, utilizes dynamic biomarker trajectories to more accurately predict progression to active multiple myeloma, potentially minimizing overtreatment for patients at low risk.
The PANGEA project, initiated in March 2021, analyzed data from a cohort of 2,344 patients with precursor conditions for multiple myeloma identified at DFCI. Researchers followed these patients longitudinally, collecting clinical and biological variables to train and validate the new risk assessment tool. The study involved a training cohort of 1,031 SMM patients and validation across five independent international cohorts, encompassing a total of 1,239 patients from institutions in Greece, the United Kingdom, Germany, Spain, and Italy.
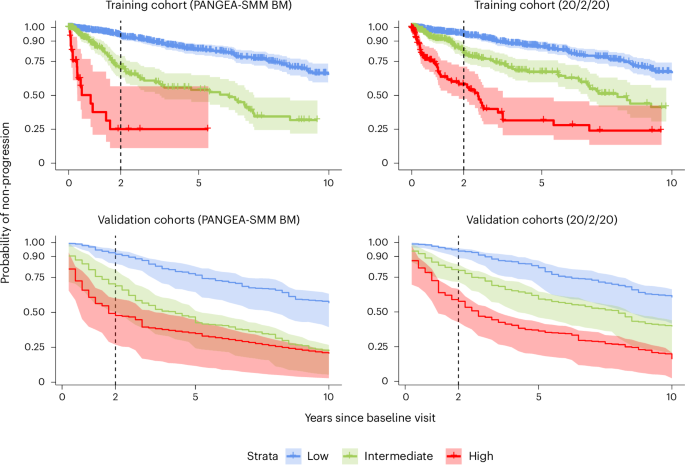
Current risk stratification models rely on static biomarkers, offering a snapshot in time. PANGEA-SMM, however, incorporates how biomarkers change over time. Four evolving biomarkers were found to be significantly associated with shorter time to progression: increases in M-protein, involved/uninvolved serum free light chain ratio, creatinine, and decreases in hemoglobin. The model assesses these changes to provide a more nuanced risk assessment.
“PANGEA-SMM outperforms established models, including the 20/2/20 and IMWG models, by more accurately predicting progression,” the study authors wrote. The model achieved a C-statistic of 0.79, even without complete biomarker history (C-statistic of 0.78) or recent bone marrow biopsy (C-statistic of 0.78). A C-statistic measures the model’s ability to discriminate between patients who will and will not progress to active myeloma.
The research team developed two versions of the PANGEA-SMM model: one incorporating bone marrow plasma cell (BMPC) data and another for use when recent BMPC information is unavailable. The models are designed to be easily implemented in clinical practice, with an open-access web application developed to facilitate risk calculation and patient stratification. The application allows clinicians to input patient data and receive a personalized risk score and classification – low, intermediate, or high – for progression to multiple myeloma.
The study was approved by the Dana-Farber/Harvard Cancer Center institutional review board, and a waiver of informed consent was granted for the retrospective data analysis due to minimal risk to patients. Informed consent was obtained from patients in some validation cohorts, adhering to ethical guidelines at each participating institution. The rigorous review process included approvals from ethics committees in Greece, the UK, Germany, Spain, and Italy.
Researchers emphasize the model’s adaptability, noting that it can function effectively even with incomplete biomarker histories by assigning a value of zero to dynamic variables when historical data is missing. Validation results remained consistent even when using less frequent observations – one per year – suggesting the model’s robustness in real-world clinical settings.
The PANGEA project is ongoing, and the open-science validation application allows researchers and clinicians to further evaluate the model’s performance on different datasets and subpopulations. The study data were collected and managed using Research Electronic Data Capture (REDCap) tools hosted at DFCI, ensuring data security and integrity.
