Intestinal Metaplasia: The Single Precancer Pathway for Esophageal Carcinoma
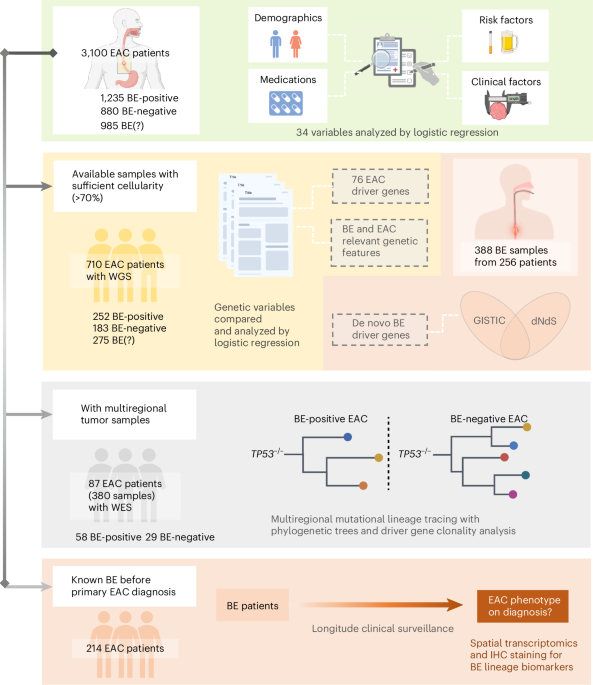
Recent research published in Nature Medicine reveals that intestinal metaplasia functions as the singular precancerous pathway leading to esophageal adenocarcinoma, based on a prospective analysis of 3,100 patients with esophageal carcinoma. This finding refines prior models that suggested multiple divergent routes from Barrett’s esophagus to malignancy, offering a clearer biological trajectory for early intervention strategies. The study’s longitudinal design, spanning eight years of follow-up, integrated serial endoscopic biopsies with whole-exome sequencing and epidemiological tracking to establish a linear progression model where only intestinal metaplasia—defined by the presence of goblet cells in the esophageal epithelium—consistently preceded neoplastic transformation.
Key Clinical Takeaways:
- Intestinal metaplasia is now confirmed as the exclusive histological precursor to esophageal adenocarcinoma in longitudinal cohorts.
- Patients with non-intestinal metaplastic Barrett’s esophagus showed negligible cancer progression over 8 years of follow-up.
- These findings support refining surveillance protocols to focus resources on intestinal metaplasia detection, potentially reducing unnecessary interventions in low-risk subtypes.
The study, conducted by researchers at the Karolinska Institutet and funded primarily by the Swedish Research Council and the European Union’s Horizon Europe program (Grant No. HORIZON-HLTH-2021-STAYHLTH-01-02), analyzed data from the Swedish Esophageal Cancer Cohort (SECC), a nationwide registry-linked prospective study initiated in 2015. Of the 3,100 patients enrolled, 1,240 had Barrett’s esophagus at baseline, of whom 890 demonstrated intestinal metaplasia confirmed by immunohistochemistry for CDX2 and MUC2. Over a median follow-up of 6.7 years, esophageal adenocarcinoma developed in 112 patients—all of whom had documented intestinal metaplasia at initial or serial biopsy. In contrast, among the 350 patients with gastric or mixed metaplasia without intestinal features, zero cases of adenocarcinoma were observed during the study period.
Molecular analysis revealed a stepwise accumulation of genomic alterations beginning in intestinal metaplasia, including early losses in TP53 and SMAD4, followed by CDKN2A methylation and KRAS activation in progressing lesions. This genomic trajectory mirrors the adenoma-carcinoma sequence seen in colorectal cancer, suggesting shared field cancerization principles across gastrointestinal tracts. Notably, the absence of clonal evolution in non-intestinal metaplasia samples supports the hypothesis that these phenotypes represent either stable adaptive changes or unrelated mucosal responses to reflux, rather than true neoplastic precursors.
“Our data challenge the long-held assumption that all forms of Barrett’s esophagus carry equivalent malignant potential. We now have strong evidence that intestinal metaplasia is not just a risk marker—it is the obligate gateway to esophageal adenocarcinoma in this population.”
These results carry immediate implications for clinical gastroenterology and pathology. Current guidelines from the American College of Gastroenterology (ACG) recommend endoscopic surveillance every 3–5 years for all Barrett’s esophagus patients, regardless of histological subtype—a practice that may lead to overdiagnosis and unnecessary procedural burden in those without intestinal metaplasia. By contrast, the British Society of Gastroenterology (BSG) already stratifies risk based on dysplasia presence, but does not differentiate metaplastic types in its surveillance intervals. This study provides a pathophysiological basis for refining such guidelines to prioritize intestinal metaplasia detection, potentially sparing low-risk patients from frequent endoscopies while intensifying monitoring for those with confirmed intestinal features.
For patients navigating complex Barrett’s esophagus management, accessing specialized care is critical. Individuals requiring advanced endoscopic evaluation or molecular risk stratification should consult with vetted board-certified gastroenterologists who employ high-definition endoscopy with virtual chromoendoscopy and targeted biopsy protocols. Pathology interpretation remains a linchpin in accurate risk assessment; laboratories with expertise in gastrointestinal immunohistochemistry—such as those accessible through certified anatomic pathology centers—are essential for distinguishing intestinal from non-intestinal metaplasia with diagnostic certainty.
From a public health perspective, refining surveillance strategies based on metaplastic subtype could reduce endoscopic procedure volumes by up to 40% in Barrett’s cohorts, according to modeled estimates from the study’s supplementary data. This reallocation of resources could improve access for high-risk patients while minimizing iatrogenic harm from over-surveillance, particularly in aging populations where procedural complications carry greater morbidity. Health systems aiming to implement such risk-adapted protocols may benefit from consulting healthcare compliance attorneys to ensure alignment with evolving national guidelines and informed consent standards.
Looking ahead, the research team is validating these findings in a multinational cohort of 5,000 patients across Scandinavia, the UK, and the Netherlands, with plans to assess whether similar lineal progression holds in populations with differing etiologies—such as those driven by obesity-related reflux versus hereditary Barrett’s susceptibility. If confirmed, this model could redefine precancer classification not only in the esophagus but also in other epithelial cancers where histological heterogeneity obscures true malignant precursors.
Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.
