Title: Medicare and Medicaid Coverage of GLP-1s: CMS Initiatives to Expand Access and Lower Costs via the BALANCE Model and Other Demonstration Programs
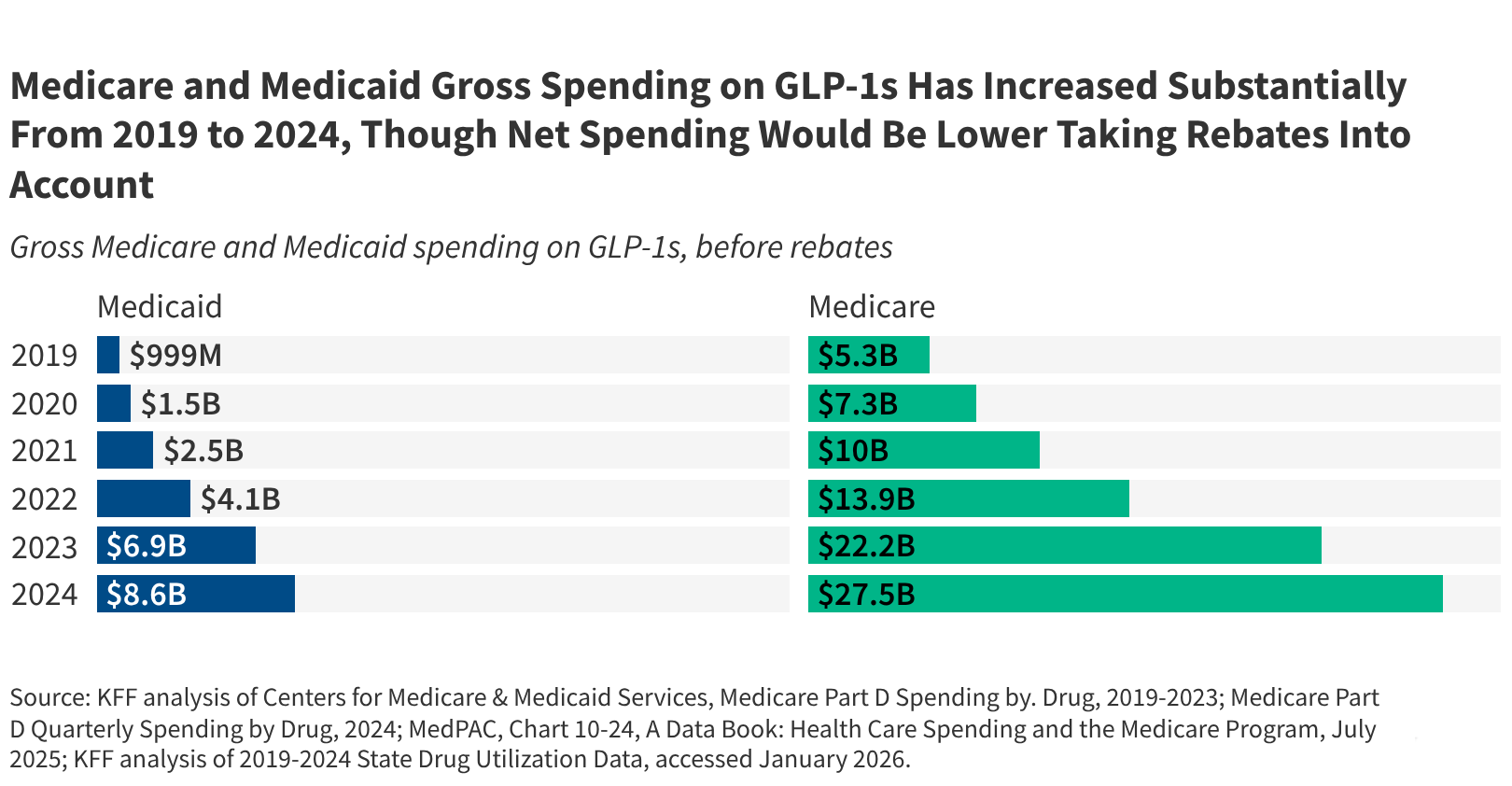
As of April 2026, the Centers for Medicare & Medicaid Services (CMS) has launched the BALANCE Model—a temporary demonstration program designed to evaluate innovative payment and delivery approaches for glucagon-like peptide-1 receptor agonists (GLP-1s) in Medicare and Medicaid populations. This initiative responds to surging demand for GLP-1 therapies like semaglutide and tirzepatide, which have demonstrated transformative efficacy in managing type 2 diabetes and obesity but remain constrained by cost barriers and uneven access, particularly among underserved beneficiaries. The BALANCE Model seeks to determine whether value-based purchasing arrangements, outcome-linked rebates, and care coordination incentives can expand equitable access while ensuring fiscal sustainability for federal healthcare programs.
- Key Clinical Takeaways:
- The BALANCE Model tests CMS’s ability to align payment with clinical outcomes for high-cost GLP-1 therapies in dual-eligible and low-income populations.
- Early pilot data suggest that integrating GLP-1s into comprehensive chronic care management reduces diabetes-related hospitalizations by up to 30% in high-risk cohorts.
- Sustainable access hinges on transparent pricing mechanisms and provider education to mitigate inappropriate prescribing and ensure adherence to evidence-based guidelines.
- The problem: Despite FDA approval and robust clinical evidence, GLP-1 receptor agonists remain underutilized in Medicare and Medicaid due to prior authorization hurdles, high out-of-pocket costs, and fragmented care delivery—particularly for beneficiaries with comorbid obesity and cardiovascular risk.
- According to a 2025 longitudinal study published in JAMA Internal Medicine, only 18% of eligible Medicare beneficiaries with type 2 diabetes and obesity received a GLP-1 prescription within one year of eligibility, compared to 42% in private insurance plans—a disparity driven largely by administrative burden and coverage restrictions (Funded by the National Institute of Diabetes and Digestive and Kidney Diseases, NIDDK Grant R01DK128765).
- GLP-1s function by enhancing glucose-dependent insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety via central nervous system pathways—mechanisms that address both hyperglycemia and adiposity pathogenesis. In the landmark SUSTAIN-6 trial (n=3,297), semaglutide reduced major adverse cardiovascular events by 26% over 2.1 years, establishing a new standard of care for cardioprotection in type 2 diabetes.
- “The real-world effectiveness of GLP-1s hinges not just on pharmacology but on systems that support long-term adherence,” says Dr. Elena Rodriguez, Professor of Endocrinology at Johns Hopkins Bloomberg School of Public Health. “Programs like BALANCE must couple medication access with behavioral support and primary care integration to avoid widening health inequities.”
“We’re seeing remarkable clinical benefits, but without structural changes in how these drugs are delivered and paid for, we risk creating a two-tiered system where only the affluent benefit.”
- Dr. Marcus Chen, Health Economist at the Kaiser Permanente Washington Health Research Institute, adds: “Value-based models like BALANCE are essential to test whether we can decouple innovation from unsustainable cost growth. If we can show that GLP-1s reduce downstream complications—like heart failure hospitalizations or dialysis initiation—then we’ve got a pathway to justify broader coverage.”
“The goal isn’t just to pay for a drug; it’s to pay for health outcomes. That’s where CMS’s demonstration authority can truly innovate.”
- For patients navigating complex coverage rules for GLP-1 therapies, consulting with board-certified endocrinologists who specialize in metabolic disease management can help optimize treatment plans and appeal prior authorizations. federally qualified health centers (FQHCs) participating in CMS innovation models often provide integrated care teams that include pharmacists and dietitians—critical for managing GLP-1 side effects like nausea and ensuring nutritional adequacy during weight loss.
- From a B2B perspective, healthcare organizations adapting to BALANCE’s outcome-based frameworks may benefit from engaging healthcare compliance attorneys experienced in federal demonstration programs to ensure accurate reporting, avoid false claims risks, and structure contracts that align with CMS’s monitoring and evaluation requirements.
- The BALANCE Model represents a pivotal test of whether federal healthcare programs can leverage innovation to improve population health without triggering budgetary unsustainability. As CMS collects data on hemoglobin A1c reduction, weight change trajectories, and healthcare utilization over the next 18 months, the findings could reshape national coverage policy for not just GLP-1s but future high-cost, high-benefit therapies in chronic disease management.
*Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.*
