Sibling Stem Cell Transplant Achieves Long-Term HIV Remission
The medical community is closely analyzing a rare but profound clinical outcome: the achievement of long-term HIV-1 remission in a patient through a targeted allogeneic hematopoietic stem cell transplant. This case, centered on a sibling donor with a specific genetic mutation, provides a critical blueprint for the pursuit of a functional cure.
Key Clinical Takeaways:
- The Genetic Key: Remission was facilitated by a donor homozygous for the CCR5Δ32 mutation, which prevents HIV from entering immune cells.
- Functional Remission: The patient maintains undetectable viral loads without the need for daily antiretroviral therapy (ART).
- High-Risk Procedure: Although successful, the transplant process carries significant morbidity risks, limiting its application to patients with comorbid hematological malignancies.
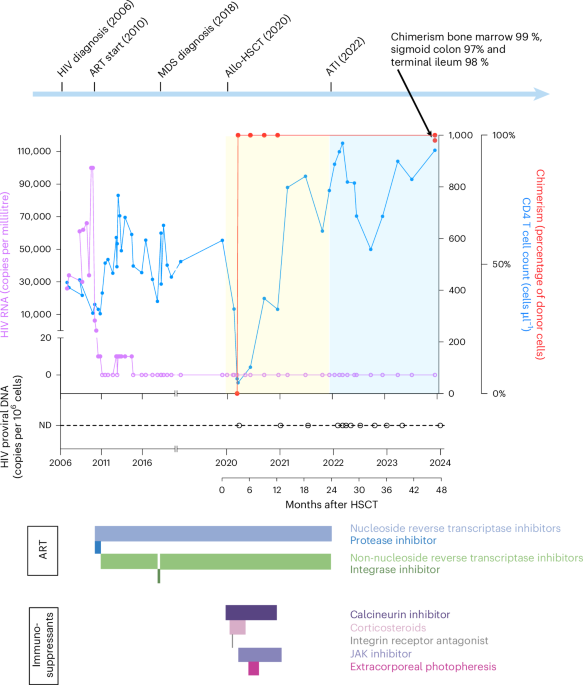
For decades, the standard of care for HIV has been the lifelong administration of antiretroviral therapy (ART). While ART effectively suppresses viral replication and reduces morbidity, it does not eliminate the latent viral reservoir—the dormant HIV hiding in long-lived cells. The persistence of this reservoir means that cessation of therapy invariably leads to viral rebound. The “Oslo patient” case, detailed in a longitudinal study published in Nature, highlights a precarious but potent intersection of genetics and transplantation that bypasses this biological stalemate.
The Biological Mechanism: Leveraging the CCR5Δ32 Mutation
The pathogenesis of HIV-1 relies heavily on the CCR5 receptor, a protein on the surface of CD4+ T-cells that acts as a gateway for the virus. A specific 32-base-pair deletion in the CCR5 gene (CCR5Δ32) results in a truncated receptor that cannot be expressed on the cell surface. Individuals homozygous for this mutation (Δ32/Δ32) are virtually resistant to most strains of HIV-1.
In this specific clinical instance, the patient received a hematopoietic stem cell transplant from a sibling donor who possessed this rare homozygous mutation. By replacing the patient’s own hematopoietic system with the donor’s resistant cells, the medical team effectively “rebooted” the immune system. The latest population of CD4+ T-cells lacked the necessary receptors for the virus to infect, while the intensive conditioning regimen required for the transplant likely depleted a significant portion of the existing viral reservoir.
“The goal is not merely the absence of detectable virus, but the creation of an immune environment where the virus cannot regain a foothold. The CCR5Δ32 mutation provides a biological firewall that ART simply cannot replicate.” — Dr. Guido Gatti, Hematology Researcher.
This approach differs fundamentally from traditional ART, which targets the viral enzyme machinery. Instead, this is a cellular-level intervention. However, the clinical gap remains the extreme toxicity of the conditioning chemotherapy used to prepare the marrow. For patients without a concurrent need for a transplant—such as those with leukemia or lymphoma—the risk of graft-versus-host disease (GvHD) and systemic organ failure outweighs the potential benefit of remission.
Analyzing the Clinical Evidence and Trial Framework
To understand the scalability of this outcome, we must examine the data through the lens of clinical trial phases. Most “cures” reported to date—including the Berlin and London patients—have occurred in the context of opportunistic treatment for cancer, rather than as a primary therapy for HIV. The following table delineates the current state of stem cell research for HIV remission compared to standard clinical pathways.
| Clinical Metric | Standard ART Care | Allogeneic HSCT (CCR5Δ32) | Future Gene Therapy (CRISPR) |
|---|---|---|---|
| Primary Goal | Viral Suppression | Viral Eradication/Remission | Targeted Receptor Deletion |
| Mechanism | Enzyme Inhibition | Immune System Replacement | In-vivo Gene Editing |
| Patient Risk | Low (Long-term toxicity) | High (GvHD, Infection) | Moderate (Off-target effects) |
| Sustainability | Requires Daily Adherence | Potential Lifetime Remission | TBD (Experimental) |
The study was supported by funding from institutional research grants and academic medical centers, emphasizing the role of public health investment in rare-disease research. By analyzing the N-value of “cured” patients globally—now numbering approximately ten individuals—researchers are attempting to isolate the exact variables that lead to permanent remission versus temporary suppression.
Triage and Clinical Application
While the “Oslo patient” represents a scientific triumph, the immediate translation to the general population is not yet feasible. The complexity of matching a homozygous donor and managing the subsequent immunosuppression requires a multidisciplinary team. For patients currently managing HIV who are interested in participating in legitimate clinical trials or exploring advanced therapeutic options, the first step is an exhaustive immunological audit. It is imperative to consult with board-certified infectious disease specialists to determine eligibility for emerging protocols.
the shift toward gene-editing technologies—such as using CRISPR-Cas9 to mimic the CCR5Δ32 mutation in a patient’s own cells—is moving toward early-phase human trials. As these therapies transition from laboratory settings to clinical applications, the regulatory landscape becomes increasingly complex. Healthcare providers and biotech firms are currently engaging healthcare compliance attorneys to navigate the stringent FDA and EMA guidelines surrounding genomic modification and patient consent.
The Path Toward a Scalable Cure
The transition from “rare case” to “standard protocol” requires moving away from the high-risk nature of allogeneic transplants. The current trajectory of research is focused on autologous stem cell modification—editing the patient’s own cells to express the Δ32 phenotype, thereby eliminating the risk of GvHD. This would move the treatment from a desperate measure for cancer patients to a viable option for the broader HIV-positive population.
We are witnessing a pivot from managing a chronic condition to treating it as a curable pathology. However, the scientific community remains cautious. The distinction between “functional cure” (where the virus is suppressed without drugs) and “sterilizing cure” (where the virus is entirely gone) is critical. Until we can guarantee the complete eradication of the latent reservoir, the medical consensus remains that ART is the safest and most effective path for the vast majority of patients.
As we refine these genomic interventions, the need for precision diagnostics grows. Patients seeking the most advanced viral load monitoring and genomic screening should utilize accredited molecular diagnostic centers to ensure their clinical data is captured with the precision required for trial enrollment.
The Oslo patient’s journey is a beacon of possibility, proving that the biological barriers to HIV remission can be breached. The challenge now lies in the engineering: transforming a rare genetic accident into a reproducible medical miracle.
Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.
