RNA Barcoding Technique Revolutionizes Brain Mapping Precision
The human brain represents the most intricate circuitry in the known universe, yet our ability to map its connections has long been hindered by the sheer scale of its complexity. A paradigm shift is currently unfolding as researchers transition from laborious physical reconstruction to high-speed molecular sequencing to decode the brain’s hidden wiring.
Key Clinical Takeaways:
- The development of Connectome-seq allows for the simultaneous mapping of thousands of neural connections with single-synapse resolution.
- Unlike previous sequencing methods that only tracked axonal reach, this new approach identifies the specific partner neurons at the synapse.
- This high-resolution mapping provides a critical foundation for understanding circuit dysfunction in neurodegenerative diseases and developing targeted therapeutic interventions.
For decades, the quest to create a comprehensive “connectome”—a complete map of neural connections—has been a grueling exercise in patience and precision. The standard of care for brain mapping traditionally involved cutting tissue into infinitesimal slices, imaging them via various microscopy techniques and manually reconstructing pathways. Even as these methods provided high detail, they lacked the scalability required to understand the systemic pathogenesis of complex neurological disorders. The clinical gap has been stark: we could see where a neuron traveled, but we rarely knew exactly which cell it was talking to at the point of contact.
The Evolution of Molecular Barcoding in Neuroscience
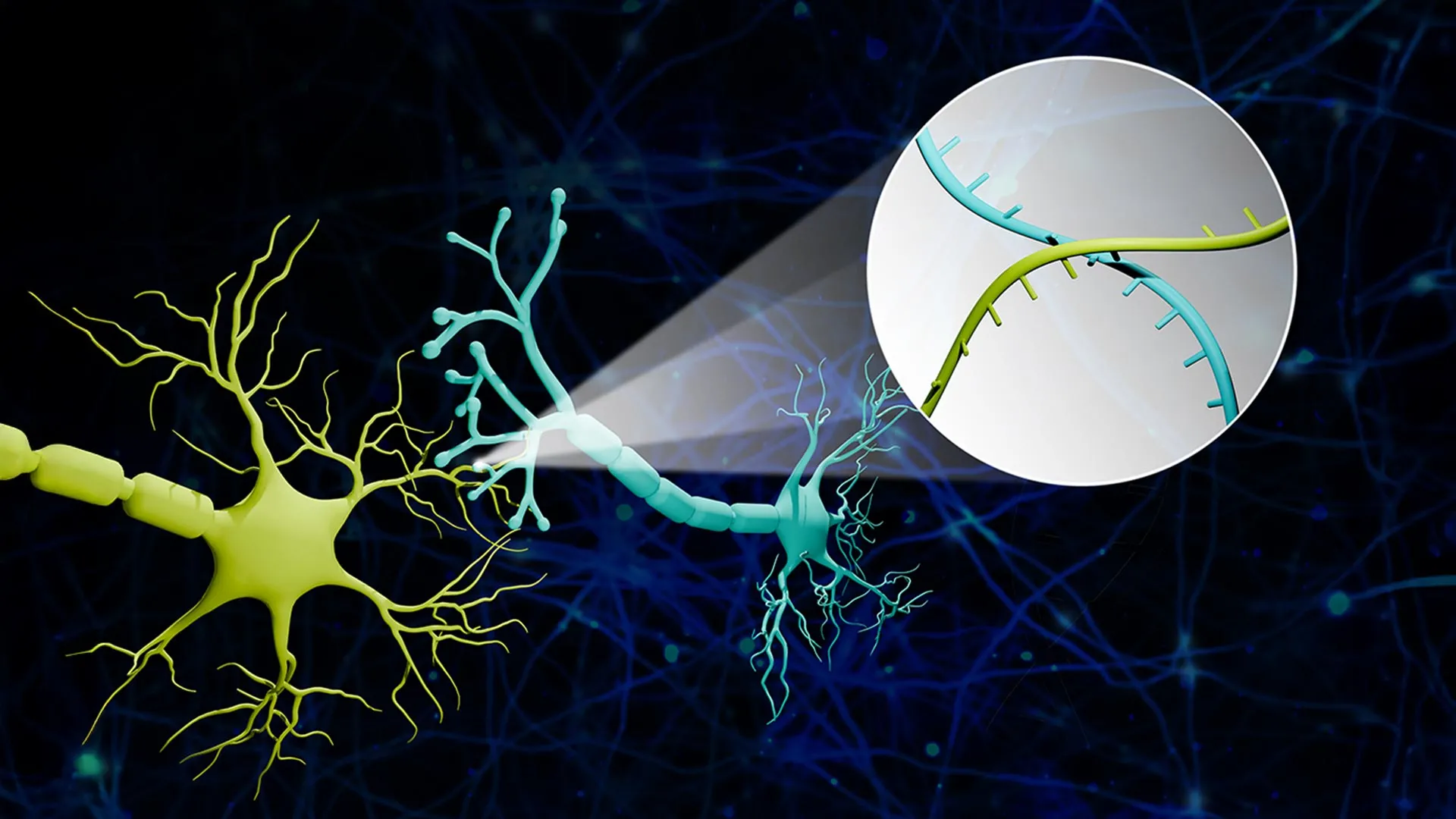
The emergence of RNA barcoding has effectively transformed brain mapping from a visual reconstruction task into a data sequencing task. By tagging individual neurons with unique molecular “barcodes,” scientists can now identify connections with unprecedented speed. This trajectory began with earlier milestones, such as the research published in Neuron, where researchers utilized random RNA sequences to map single neuron projections. However, early iterations of this technology often lacked the granularity to pinpoint the exact synaptic partner, providing a general map of reach rather than a precise map of connection.
The latest breakthrough, developed by a team led by Boxuan Zhao, a professor of cell and developmental biology at the University of Illinois Urbana-Champaign, addresses this limitation through a platform called Connectome-seq. Published in Nature Methods, this technology achieves single-synapse resolution. By utilizing RNA barcodes to tag neurons, the team can map thousands of connections simultaneously, bypassing the necessitate for the slow, slice-by-slice imaging of the past.
“When engineering a computer, you need to know the circuitry of the central processing unit. If you don’t know how everything is wired together, you can’t understand its function, optimize it or fix it when something breaks. We are approaching the brain the same way,” stated study leader Boxuan Zhao.
This level of precision is not merely an academic achievement; it is a clinical necessity. For patients suffering from cognitive decline or motor dysfunction, the underlying issue is often not the death of a single cell, but the collapse of a specific circuit. Identifying these broken links requires the exact resolution that Connectome-seq provides. When these circuit failures manifest as chronic symptoms, patients often require the expertise of board-certified neurologists to manage the resulting morbidity and coordinate long-term care.
Integrating Transcriptomes with Synaptic Architecture
Parallel to the operate at the University of Illinois, researchers at the Broad Institute of Harvard and MIT have explored the intersection of genetic expression and physical connectivity. Their work on protein-guided RNA barcoding, detailed in a bioRxiv preprint, utilizes a method called Synapse-seq. In studies focusing on the mouse primary visual cortex, this approach successfully recovered known long-range projections and uncovered previously unknown cortical layer connections.
By linking the transcriptome—the complete set of RNA transcripts produced by the genome—to the synaptic architecture, scientists can now observe how the genetic identity of a neuron influences who it connects with and how that connection functions. This synthesis of genetic and structural data is pivotal for understanding the etiology of neurodegenerative diseases. When a protein misfolds or a gene is silenced, the resulting structural degradation of the synapse can be tracked in real-time through these barcoding techniques.
The ability to map these connections in mice provides a scalable model for human application. As we move toward a deeper understanding of how these circuits fail, the demand for high-precision diagnostics will grow. Facilities capable of advanced neuroimaging and genetic screening, such as specialized neurological diagnostic centers, will be essential in translating these research breakthroughs into patient-side diagnostics.
Clinical Implications for Neurodegenerative Disease
The primary clinical value of Connectome-seq and Synapse-seq lies in their application to circuit-guided therapeutic interventions. Most current treatments for neurodegenerative conditions are systemic or focused on general neurotransmitter levels. However, if a specific circuit responsible for memory or motor control is the primary site of dysfunction, a more targeted approach is required.

By identifying the exact neurons involved in a dysfunctional circuit, clinicians can theoretically develop interventions that target only the affected pathways, reducing off-target effects and increasing efficacy. This shift from “global” treatment to “circuit-specific” treatment represents the next frontier of precision medicine in neurology. For healthcare providers and pharmaceutical developers, navigating the regulatory hurdles of such targeted therapies often requires the guidance of healthcare compliance attorneys to ensure that new protocols meet evolving safety and ethical standards.
The speed of this new mapping technology also allows for a more rapid study of disease progression. Rather than spending years mapping a single region of the brain, researchers can now observe how connections erode over the course of a disease in a fraction of the time. This acceleration is critical for the development of early-detection biomarkers, which could allow for intervention years before the onset of severe clinical symptoms.
As we refine these molecular tools, the transition from mouse models to human clinical application will likely depend on our ability to integrate this data into existing healthcare frameworks. The journey from a sequenced RNA barcode to a bedside treatment is complex, but the roadmap is becoming clearer. The future of neurology lies in our ability to treat the brain not as a collection of cells, but as a precise, addressable network of connections.
For those seeking to understand how these emerging neurological insights apply to their own health or the health of a loved one, consulting with vetted specialists remains the gold standard. Finding a provider who stays current with peer-reviewed advancements in synaptic research is the first step toward accessing the next generation of neurological care.
Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.
