New Method Decouples Alzheimer’s Disease Pathology on PET Scans
The diagnostic landscape for neurodegenerative diseases is shifting from broad clinical observation to molecular precision. For decades, the overlap in symptoms between various forms of dementia has left clinicians relying on a process of elimination. Yet, the integration of advanced PET imaging is now allowing the medical community to separate, or decouple, the specific pathological drivers of cognitive decline.
Key Clinical Takeaways:
- Combined Amyloid-PET and FDG-PET scans provide a high-accuracy predictive model for the progression of amnestic mild cognitive impairment (MCI) to full-scale dementia.
- Tau neurofibrillary tangles and beta-amyloid deposits act as primary drivers of Alzheimer’s symptoms, with evidence suggesting women may experience faster cognitive decline.
- Advanced PET and MRI modalities are now capable of detecting LATE (limbic-predominant age-related TDP-43 encephalopathy), a condition frequently misdiagnosed as Alzheimer’s disease.
The primary clinical gap in dementia care has long been the “diagnostic shadow”—the tendency for one pathology to mask another. Many patients present with amnestic mild cognitive impairment (MCI), a state of cognitive decline that is more pronounced than expected for an individual’s age but does not yet meet the full criteria for dementia. The critical challenge for healthcare providers is determining which of these patients will progress to dementia and which are experiencing a stable or benign form of cognitive aging. Without precise biomarkers, the risk of initiating inappropriate pharmacological interventions is high.
The Synergy of Combined PET Modalities
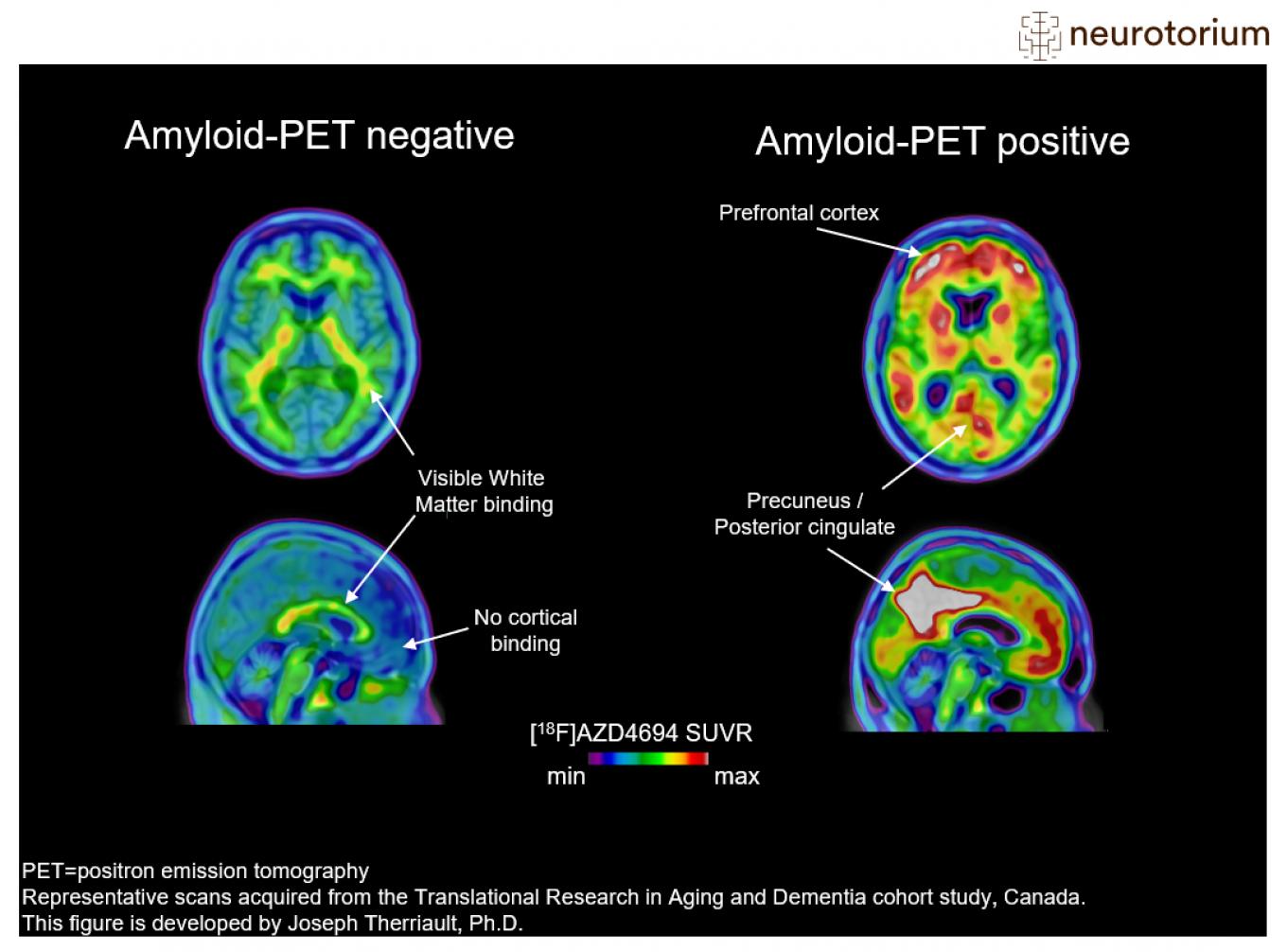
The current standard of care is evolving toward a multi-modal imaging approach. Even as a single PET scan provides a snapshot of a specific protein or metabolic state, combining Amyloid-PET with FDG-PET creates a comprehensive map of both pathology and function. Amyloid-PET identifies the presence of beta-amyloid plaques, the hallmark proteinopathy of Alzheimer’s. In contrast, FDG-PET measures glucose metabolism, highlighting areas of the brain that are no longer functioning efficiently.

Clinical data indicates that patients with amnestic MCI who exhibit both a positive Amyloid-PET scan and a characteristic Alzheimer’s pattern on FDG-PET are significantly more likely to progress to dementia. This combined approach allows clinicians to decouple the mere presence of plaques from the actual functional decline of the brain. For patients in this precarious transitional phase, early intervention is vital. It is highly recommended that individuals experiencing early memory loss consult with board-certified neurologists to establish a baseline and determine the necessity of advanced imaging.
Tau Drivers and Gender-Based Cognitive Divergence
While beta-amyloid deposits often appear early, the build-up of tau neurofibrillary tangles is recognized as a more direct driver of the clinical symptoms associated with Alzheimer’s disease. The pathogenesis involves the collapse of microtubules within neurons, leading to a breakdown in nutrient transport and eventual cell death. This biological mechanism does not affect all populations equally.
The build-up of tau neurofibrillary tangles in the brain is a key driver of Alzheimer’s disease symptoms, along with beta-amyloid deposits, and PET imaging suggests these drivers may predict faster cognitive decline in women than in men.
This divergence suggests that the morbidity associated with tau pathology may be modulated by biological sex, requiring a more nuanced approach to prognosis and treatment planning. Understanding these disparities is essential for developing personalized care pathways. To ensure the most accurate interpretation of these complex biomarkers, patients and providers should utilize specialized diagnostic imaging centers equipped with the latest PET tracers and quantitative analysis software.
Decoupling Alzheimer’s from LATE
One of the most significant breakthroughs in molecular imaging is the ability to distinguish Alzheimer’s from limbic-predominant age-related TDP-43 encephalopathy, or LATE. Because LATE targets the limbic system—the area of the brain responsible for memory and emotion—it frequently mimics the clinical presentation of Alzheimer’s. This “mimicry” has historically led to widespread misdiagnosis in elderly populations.
The apply of combined PET and MRI allows for the detection of the specific proteinopathy associated with LATE, effectively decoupling it from the amyloid-driven pathology of Alzheimer’s. This distinction is not merely academic; it is clinically imperative. The treatment protocols for Alzheimer’s are designed to target amyloid and tau, whereas LATE involves the TDP-43 protein. Applying an Alzheimer’s-specific treatment to a patient with LATE is unlikely to yield therapeutic benefits and may expose the patient to unnecessary side effects.
Managing these complex, overlapping dementias requires a multidisciplinary approach. Families navigating these diagnoses are encouraged to seek guidance from geriatric neurology specialists who can integrate imaging results with comprehensive clinical assessments.
The Future of Molecular Diagnostics
The shift toward decoupling pathologies via PET and MRI represents a move toward a “precision neurology” model. By isolating the specific protein signatures—whether they be beta-amyloid, tau, or TDP-43—clinicians can move away from the broad label of “dementia” and toward a molecularly defined diagnosis. This evolution is critical for the success of emerging disease-modifying therapies, which require highly specific patient populations to demonstrate efficacy.
As these imaging techniques become more integrated into the standard of care, the focus will likely shift toward the pre-symptomatic phase, identifying protein accumulation years before the first sign of memory loss. The ability to predict progression with high statistical probability allows for a window of opportunity in which lifestyle interventions and pharmacological trials may have the greatest impact. For those seeking the highest standard of neurodegenerative care, accessing vetted specialists through our directory ensures a connection with providers at the forefront of this molecular revolution.
Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.
