Brain Lesions and Unexplained Symptoms: Doctors Discover New Parasite in Humans
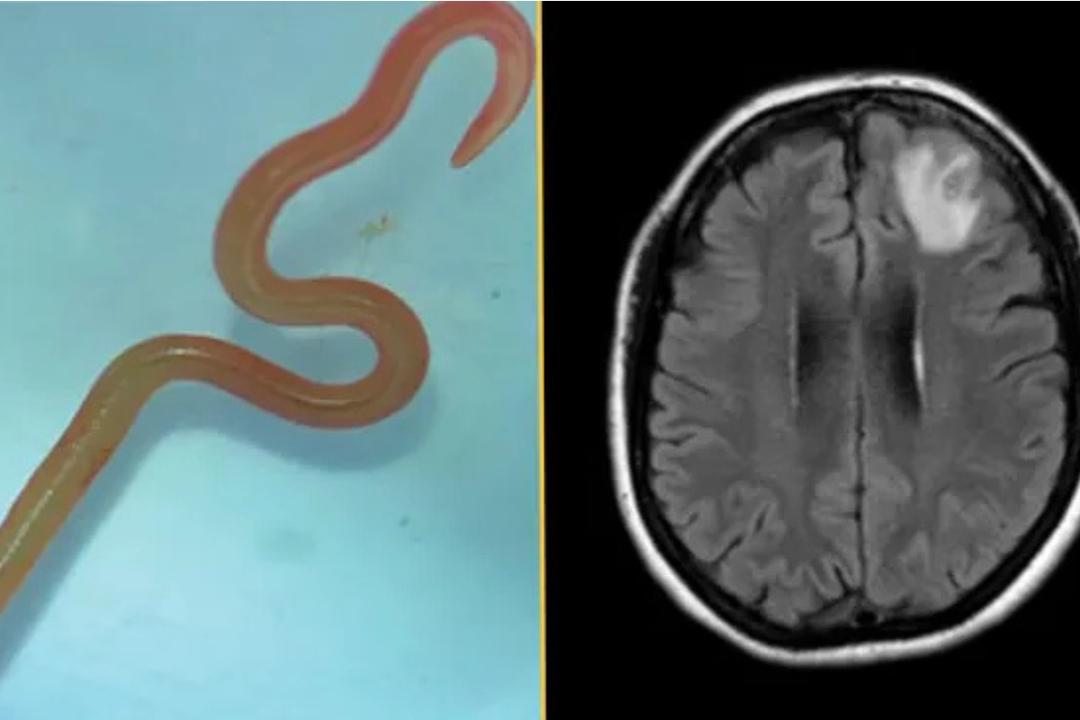
In April 2026, French researchers reported the first documented case of a novel parasitic infection in humans causing unexplained cerebral lesions and neurological symptoms, marking a significant development in neuroinfectious disease surveillance. The case, identified at Pitié-Salpêtrière Hospital in Paris, involved a 34-year-old patient presenting with progressive headaches, transient visual disturbances, and mild cognitive impairment unresponsive to standard diagnostic workup. Advanced neuroimaging revealed multiple non-enhancing cerebral lesions, prompting further investigation that ultimately led to the identification of a previously undescribed eukaryotic microorganism via cerebrospinal fluid metagenomic sequencing. This discovery underscores the evolving landscape of zoonotic pathogens and highlights critical gaps in clinical recognition of atypical infectious etiologies behind idiopathic neurological syndromes.
Key Clinical Takeaways:
- A novel parasite was detected in human cerebrospinal fluid using unbiased metagenomic sequencing, representing the first known human infection by this organism.
- The patient exhibited subacute neurological symptoms and MRI-defined cerebral lesions without evidence of autoimmune or neoplastic etiology.
- No approved antiparasitic regimen exists for this pathogen; treatment was empiric and guided by expert consultation, emphasizing the need for specialized diagnostic and therapeutic pathways.
The index case emerged during an investigative workup for suspected autoimmune encephalitis after initial serologies, CSF analysis for common pathogens (including HSV, VZV, enteroviruses, and Mycobacterium tuberculosis), and paraneoplastic panels returned negative. Despite immunotherapy, symptoms progressed, prompting clinicians to pursue broader molecular diagnostics. Metagenomic next-generation sequencing (mNGS) of CSF revealed sequences with low homology to known Protista, later validated through PCR and electron microscopy as a free-living amoeboid organism tentatively classified within the Heterolobosea clade. Phylogenetic analysis suggested closest environmental relatives found in freshwater biofilms, raising questions about potential exposure routes such as aerosolized water or nasal irrigation practices—a transmission vector epidemiologically linked to other rare CNS infections like Naegleria fowleri.
According to the study published in Emerging Infectious Diseases (April 2026), the patient had no history of immunosuppression, international travel to endemic zones, or freshwater exposure, complicating source attribution. Yet, epidemiological investigations identified potential biofilm exposure during routine household plumbing maintenance. The organism demonstrated thermotolerance up to 42°C in vitro and exhibited cytopathic effects on human neuronal cultures, suggesting direct neurotoxic potential beyond inflammatory-mediated damage. Histopathology from a subsequent biopsy (performed due to diagnostic uncertainty) showed perivascular lymphocytic infiltration and microglial activation, consistent with meningoencephalitis, though no cysts or trophozoites were visualized in tissue—likely due to low parasitic burden or focal distribution.
“This case exemplifies why mNGS should be considered earlier in the diagnostic algorithm for unexplained meningoencephalitis, particularly when standard testing fails. We’re likely underdetecting these pathogens due to lack of clinical suspicion and limited assay availability.”
The diagnostic odyssey faced significant delays due to limited awareness of emerging free-living amoebae beyond the well-known pathogens (Acanthamoeba, Balamuthia mandrillaris, Naegleria fowleri). Unlike those agents, this organism does not thrive on standard agar cultures, necessitating molecular methods for detection—a barrier in many clinical laboratories. Funding for the investigation was provided by the French National Research Agency (ANR) under its “Emerging Pathogens” initiative (Grant ANR-22-PATH-0004), with sequencing performed at the Pathogen Discovery Unit of Sorbonne Université. No pharmaceutical industry sponsorship was involved, preserving analytical independence.
Clinically, management remains undefined. The patient received a combination of miltefosine and azithromycin based on in vitro susceptibility data and extrapolation from Balamuthia treatment protocols, followed by clinical and radiographic stabilization over six weeks. However, relapse risk, long-term neurological sequelae, and optimal duration of therapy remain unknown. There are currently no FDA- or EMA-approved antiparasitic agents with proven efficacy against this specific organism, and no clinical trials are registered. This therapeutic vacuum places significant burden on neuroinfectious disease specialists and reference laboratories to guide off-label use based on mechanistic plausibility rather than robust efficacy data.
For patients presenting with unexplained neurological symptoms and atypical imaging findings, early involvement of neuroimmunology and infectious disease specialists is critical. Institutions with advanced molecular diagnostics capabilities—such as university-affiliated medical centers—are best positioned to detect such elusive pathogens. It is highly recommended to consult with vetted board-certified neurologists or infectious disease specialists experienced in neuroinflammatory disorders to guide diagnostic escalation. Facilities requiring validation of novel molecular assays may benefit from engaging CLIA-certified diagnostic laboratories with expertise in metagenomic sequencing to ensure accurate and timely pathogen identification.
This case reinforces the necessity of integrating genomic surveillance into clinical neurology workflows, particularly as environmental changes and human behavior alter exposure risks to opportunistic microbes. While the public health threat appears limited at present—no secondary cases have been identified—the incident serves as a sentinel event urging heightened vigilance among clinicians evaluating cryptogenic encephalitis. As global diagnostics advance, we must balance innovation with restraint, avoiding overtesting while ensuring that rare but treatable etiologies are not missed due to diagnostic nihilism.
“One case does not an outbreak develop, but it does reveal a diagnostic blind spot. We need standardized reporting frameworks for novel CNS pathogens to distinguish true emergence from improved detection.”
Moving forward, prospective surveillance studies are warranted to determine the true prevalence of this organism in environmental reservoirs and asymptomatic human populations. Development of specific serological or antigen detection assays could facilitate larger epidemiological investigations. Until then, clinicians must maintain a broad differential for encephalitis syndromes, leveraging advanced diagnostics judiciously and collaborating with specialized centers equipped to manage diagnostic uncertainty. The convergence of clinical acumen, technological innovation, and interdisciplinary collaboration remains our strongest defense against the evolving threat of unknown neurotropic pathogens.
*Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.*
