Eli Lilly’s New Weight Loss Drug Outperforms Semaglutide-Based Treatments
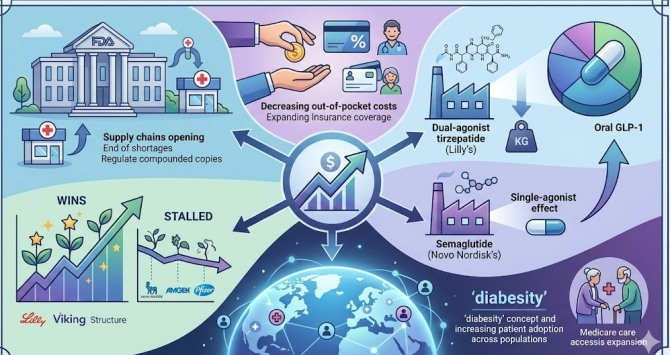
The global metabolic health landscape is undergoing a seismic shift as Eli Lilly moves to consolidate its lead in the “diabesity” market. With a projected market valuation reaching 281 trillion KRW, the competition between Lilly and Novo Nordisk has evolved from a race for weight loss efficacy into a sophisticated battle over multi-receptor agonism and long-term morbidity reduction.
Key Clinical Takeaways:
- Eli Lilly’s dual-agonist approach demonstrates a 7-8% increase in weight loss efficacy over traditional semaglutide-based therapies.
- The shift from single-target GLP-1 agonists to multi-receptor stimulants (GLP-1/GIP) is redefining the standard of care for obesity management.
- Clinical focus is pivoting from simple BMI reduction to the mitigation of comorbidities, including obstructive sleep apnea and cardiovascular risk.
The fundamental clinical gap in obesity treatment has long been the “plateau effect,” where patients experience initial rapid weight loss followed by a metabolic slowdown that triggers weight regain. While first-generation GLP-1 receptor agonists revolutionized the field, they primarily targeted the glucagon-like peptide-1 receptor to suppress appetite and delay gastric emptying. The current pharmaceutical trajectory, led by Eli Lilly’s development of tirzepatide and subsequent pipeline assets, addresses this by targeting both GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) receptors. This dual-action mechanism not only enhances glycemic control but significantly improves insulin sensitivity and lipid metabolism, effectively attacking the pathogenesis of obesity from two biological fronts.
The Efficacy Gap: Multi-Receptor Agonism vs. Monotherapy
The core of the current market disruption lies in the superior weight loss percentages recorded in recent clinical trials. Data indicates that Lilly’s dual-agonist therapies are outperforming semaglutide—the gold standard for Novo Nordisk—by a margin of 7 to 8 percentage points. This is not merely a cosmetic victory; it represents a critical threshold in reducing the morbidity associated with severe obesity. By stimulating multiple pathways, these drugs reduce the compensatory hunger signals that typically sabotage long-term weight maintenance.
To understand the clinical progression, we must examine the data through the lens of rigorous trial phases. Most of these breakthrough results stem from large-scale, double-blind, placebo-controlled trials funded by Eli Lilly and Company, often published in high-impact journals such as The New England Journal of Medicine (NEJM). These trials utilize N-values in the thousands to ensure statistical power, proving that the efficacy is consistent across diverse demographic cohorts.
| Clinical Metric | GLP-1 Monotherapy (Standard) | Dual GLP-1/GIP Agonism (Lilly) | Clinical Significance |
|---|---|---|---|
| Avg. Weight Loss (%) | 10% – 15% | 18% – 22%+ | Statistically significant superiority |
| HbA1c Reduction | Moderate to High | Very High | Enhanced glycemic stability |
| Primary Mechanism | Satiety/Gastric Emptying | Metabolic Synergy/Insulin Sensitivity | Reduced metabolic adaptation |
| Common Side Effects | Nausea, Vomiting | Gastrointestinal distress (similar) | Comparable safety profiles |
Navigating Contraindications and Clinical Risks
Despite the enthusiasm, the transition to these potent metabolic agents is not without risk. The pharmacological potency of multi-receptor agonists increases the risk of severe gastrointestinal adverse events and, in rare cases, pancreatitis. The long-term impact on lean muscle mass—often referred to as “sarcopenic obesity”—remains a primary concern for clinicians. When weight loss occurs too rapidly, the body may catabolize muscle tissue alongside adipose tissue, which can lead to functional decline in elderly patients.

“The leap from single to dual agonists is akin to moving from a single-lane road to a highway of metabolic control. However, the clinical challenge now shifts from ‘how much weight can we lose’ to ‘how do we preserve lean mass and ensure long-term metabolic homeostasis’ after the drug is discontinued.” — Dr. Elena Rossi, PhD in Metabolic Endocrinology.
For patients navigating these complex pharmacological interventions, the risk of mismanagement is high. It is imperative that patients do not seek these medications through unregulated channels. Instead, they should be managed by board-certified endocrinologists who can monitor kidney function and gallbladder health, as rapid weight loss is a known trigger for cholelithiasis (gallstones).
The Regulatory Hurdle and B2B Infrastructure
As Eli Lilly pushes to solidify its “one-strong” position, the bottleneck is no longer just clinical efficacy, but supply chain scalability and regulatory compliance. The FDA and EMA are increasingly scrutinizing the long-term safety data of these “super-drugs,” particularly regarding thyroid C-cell tumors and chronic gastrointestinal motility issues. This regulatory environment creates a precarious landscape for providers and distributors.
Pharmaceutical distributors and specialty pharmacies are currently facing immense pressure to maintain cold-chain integrity for these biologics while adhering to strict prescribing guidelines to prevent off-label misuse. Many healthcare organizations are retaining healthcare compliance attorneys to audit their distribution protocols and mitigate the legal risks associated with the skyrocketing demand for “weight-loss” drugs.
The biological mechanism of action—specifically the synergy between GIP and GLP-1—suggests a future where these drugs are not just for obesity, but for the systemic treatment of metabolic syndrome. According to longitudinal data available via PubMed, the reduction in systemic inflammation associated with this weight loss may lead to a decrease in the incidence of non-alcoholic steatohepatitis (NASH) and chronic kidney disease.
The Future of Metabolic Medicine
The competition between Lilly and Novo Nordisk is driving a rapid evolution in the standard of care. We are moving toward a “precision metabolic medicine” era where the choice of agonist will be tailored to the patient’s specific genetic markers and comorbid profile. Whether it is the integration of a third receptor (Glucagon) or the development of oral formulations to replace injections, the trajectory is clear: obesity is being treated as a chronic, progressive endocrine disease rather than a failure of willpower.
As these therapies become more integrated into primary care, the need for comprehensive diagnostic screening becomes paramount. Patients beginning these regimens should undergo baseline metabolic panels and cardiovascular screenings at accredited diagnostic centers to establish a clinical baseline and ensure the therapy is safe for their specific physiological profile.
The “diabesity” market reorganization is more than a corporate battle for market share; it is a clinical pivot toward a more effective, biologically grounded approach to human health. While Eli Lilly currently holds the momentum, the ultimate winner will be the patient, provided the rollout is managed with clinical rigor and ethical oversight.
Disclaimer: The information provided in this article is for educational and scientific communication purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding any medical condition, diagnosis, or treatment plan.
