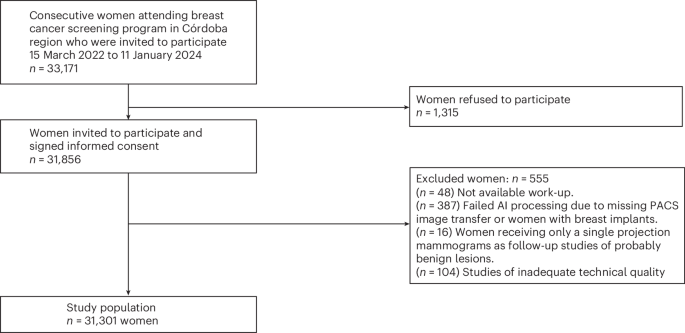
A new clinical trial in Spain has demonstrated that artificial intelligence (AI) can significantly reduce the workload for radiologists screening mammograms whereas maintaining, and in some cases improving, cancer detection rates. The study, conducted at the Córdoba Breast Cancer Screening Unit, involved over 31,000 women between March 2022 and January 2024.
Researchers evaluated a partially automated AI workflow where cases classified as low risk by the AI system, Transpara (version 1.7 ScreenPoint Medical), were automatically considered normal, bypassing the necessitate for double reading by human radiologists. The remaining cases, flagged as potentially cancerous, were then double-read with AI support. This approach resulted in a 63.6% reduction in radiologist workload, according to findings published in Nature.
The cancer detection rate increased from 6.3 per 1,000 screenings to 7.3 per 1,000 screenings – a 15.2% increase (95% confidence interval 6.6%, 24.4%; P < 0.001). However, the recall rate also increased by 14.8% (95% confidence interval 9.0%, 20.6%).
The trial, registered as NCT04849776 and NCT04949776 on ClinicalTrials.gov, utilized both digital mammography (DM) and digital breast tomosynthesis (DBT). Workload reduction was observed in both modalities – 62.1% for DM, and 65.5% for DBT. The impact on cancer detection and recall rates differed slightly between the two. In DM, the cancer detection rate increased by 1.6 per 1,000 and the recall rate by 1.3%, while both rates remained stable in DBT.
The AI system, Transpara, has been the subject of prior research, with previous studies demonstrating its ability to achieve breast cancer detection performance comparable to radiologists and improve accuracy when used as a decision support tool. The system analyzes mammograms using deep convolutional neural networks, having been trained on a database of over 15 million images from across North America, Europe, and Asia, including over 15,000 malignant cases.
Participants in the study, women aged 50 to 71, provided written informed consent. The study protocol received a favorable ruling from the Institutional Review Board (IRB) at Reina Sofía University Hospital of Córdoba Research Ethics Committee (IRB No 4932) in March 2021. The study was conducted as part of the Andalusian screening program in Spain.
Researchers emphasized that the human readers in the AI strategy ultimately made the decision to recall a woman or not, despite having access to the AI system’s analysis and risk scores. No adverse events were reported during the trial. The individual deidentified participant dataset, data dictionary, and study protocol are publicly accessible via Zenodo at https://doi.org/10.5281/zenodo.17625633.
Leave a Reply