A bacterium commonly found in the human mouth, Fusobacterium nucleatum, has been linked to the growth and spread of breast cancer, according to a new study from the Johns Hopkins Kimmel Cancer Center and the Bloomberg~Kimmel Institute for Cancer Immunotherapy. The research, published in Cell Communication and Signaling, suggests the oral microbe can reside in breast tissue and contribute to tumor development by damaging DNA and altering cellular behavior.
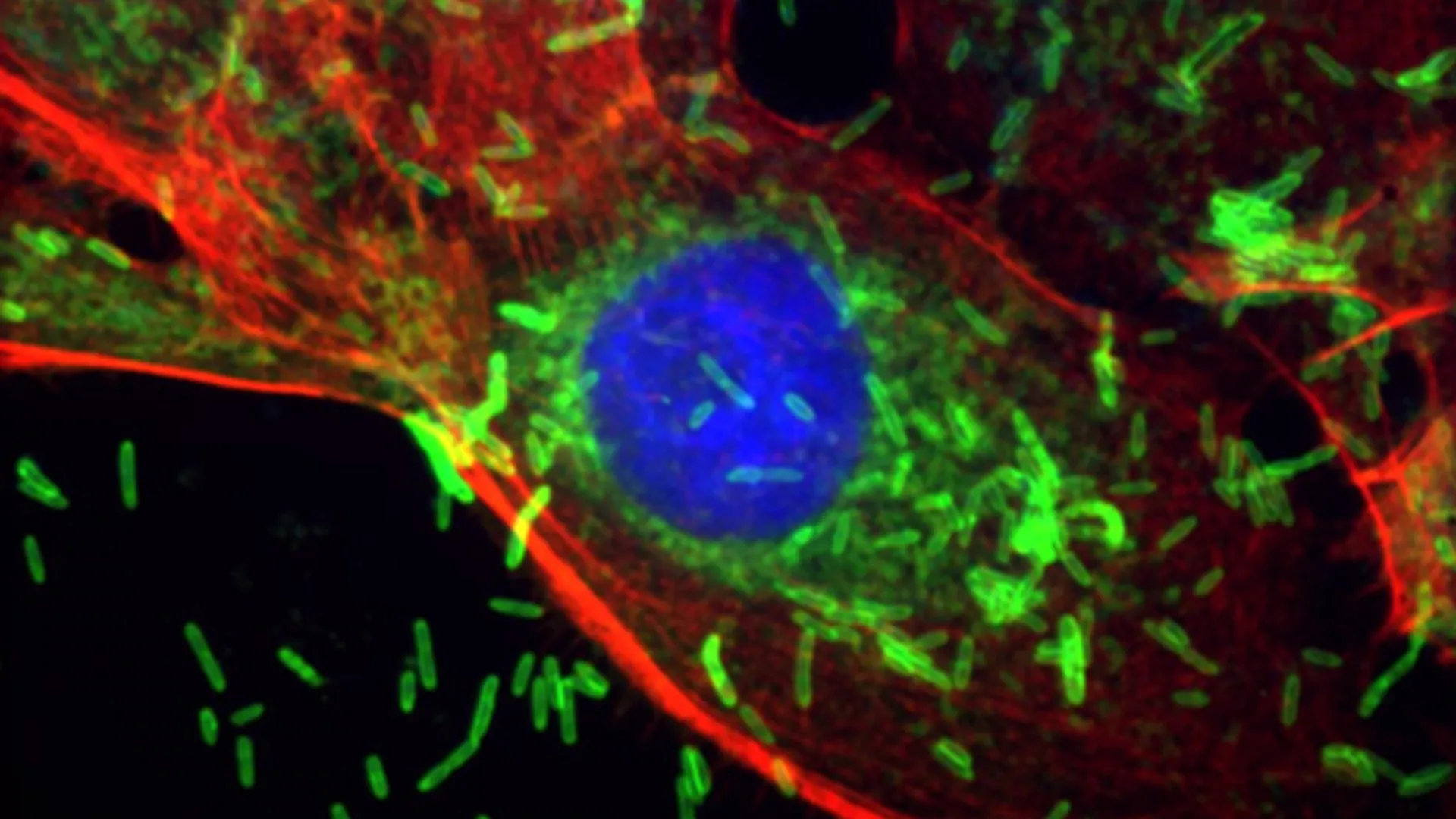
Researchers found that F. Nucleatum, previously associated with colorectal and other cancers, can enter the bloodstream and establish itself within breast tissue. Once present, the bacterium triggers inflammation and initiates cellular changes often seen in the early stages of cancer development. Experiments conducted on animal models of human breast cancer demonstrated that the presence of F. Nucleatum accelerated tumor growth and increased the metastasis of cancer cells to the lungs.
“The key takeaway is that this oral microbe can reside in breast tissue and that there is a connection between this pathogen and breast cancer,” said Dr. Sharma, a researcher involved in the study. The research was prompted by numerous smaller studies that had previously indicated a correlation between periodontal disease and an increased risk of breast cancer, according to the team.
The study details how F. Nucleatum impacts tissue at a cellular level. Introducing the bacterium directly into the breast ducts of mice resulted in metaplastic and hyperplastic lesions – noncancerous changes characterized by excessive cell growth or alterations in cell type. These changes were accompanied by inflammation, DNA damage, and increased cell proliferation. When the bacterium entered the bloodstream, it significantly enhanced the growth and spread of existing tumors.
A critical mechanism identified by the researchers involves DNA damage and the activation of cellular repair systems. Exposure to F. Nucleatum caused DNA damage, prompting the activation of repair mechanisms, specifically nonhomologous end joining. While this process rapidly reconnects broken DNA strands, it is prone to introducing mutations. The study found that even brief exposure to the bacterium increased levels of a protein called PKcs, which is associated with increased cancer cell movement, invasion, stem-like characteristics, and resistance to chemotherapy.
Certain cells exhibited a heightened susceptibility to the effects of F. Nucleatum. Epithelial cells, which line the breast ducts, and breast cancer cells carrying BRCA1 mutations were particularly vulnerable. BRCA1-mutant cells displayed elevated levels of a surface sugar, Gal-GalNAc, which facilitates bacterial attachment and entry into cells. These cells absorbed more F. Nucleatum and retained it over multiple cell generations, intensifying DNA damage and promoting cancer development.
“Our findings reveal a link between oral microbes and breast cancer risk and progression, particularly in genetically susceptible individuals,” Dr. Sharma explained. “Nothing happens in isolation. The results suggest that multiple risk factors come together with F. Nucleatum acting as an environmental factor that may cooperate with inherited BRCA1 mutations to promote breast cancer and tumor aggressiveness.”
The research team, including Sheetal Parida, Deeptashree Nandi, Deepak Verma, Mingyang Yi, Ashutosh Yendi, Jessica Queen, Kathleen Gabrielson and Cynthia Sears, emphasized the require for further investigation to determine how these findings can be applied to patient care. Future studies will focus on exploring whether improved oral health practices could potentially reduce breast cancer risk.
The study was funded by grants from the Breast Cancer Research Foundation, Congressionally Directed Medical Research Programs Department of Defense Breast Cancer Research Program (BC191572 and BC210668), the John Fetting Fund for Breast Cancer Prevention and the Bloomberg~Kimmel Institute for Cancer Immunotherapy.
Leave a Reply