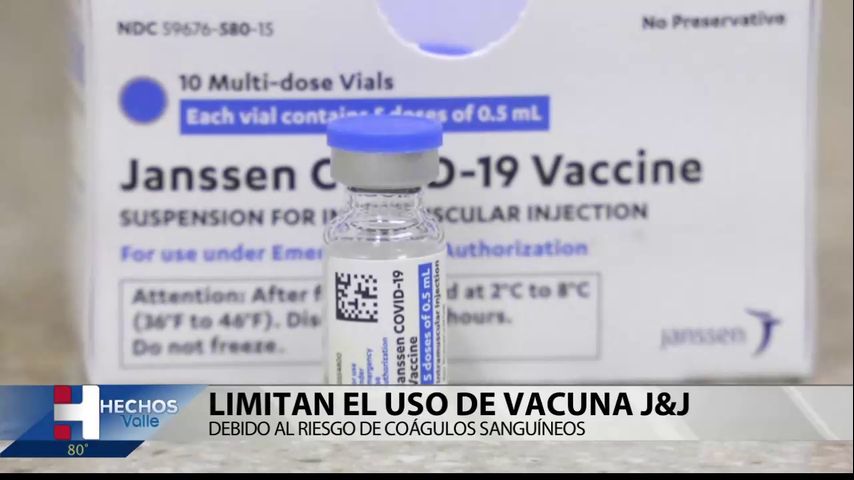
Limit use of J&J vaccine due to risk of blood clots
The FDA announced that it will limit the emergency use authorization of Johnson & Johnson’s COVID-19 vaccine to people 18 years of age and older. In an FDA statement, it was reported that the change is motivated by the risk of a rare and dangerous clotting condition called thrombosis with thrombocytopenia syndrome, which can occur … Read more